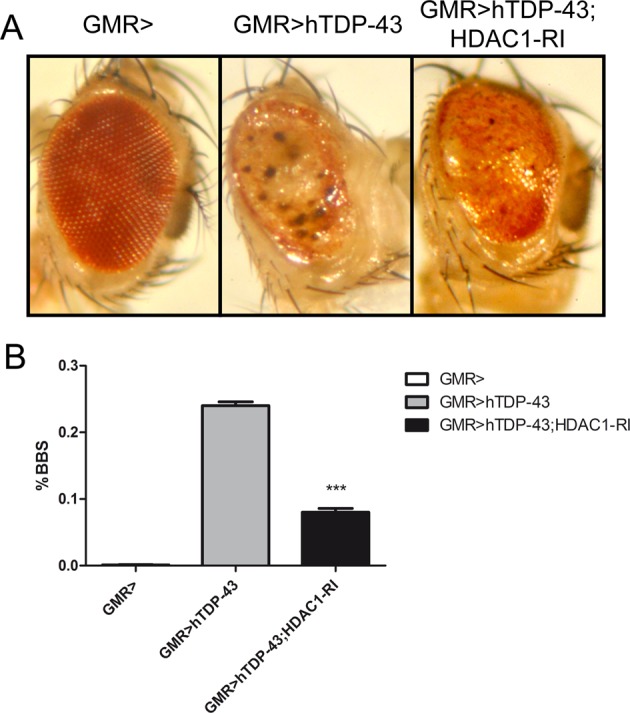
TDP-43 pathology is characteristic that characterizes the disease amyotrophic lateral sclerosis (ALS) and frontotemporal lobar degeneration (FTLD-TDP). TDP-43 experience some posttranslational modifications that can alter the biological activity and tendency to aggregate, which is a common characteristic of the different neurodegenerative conditions.
new evidence provided by the present study refers to the TDP-43 acetylation in cellular models of ALS. Using both in vitro and in vivo approaches, we show that the TDP-43 interact with histone deacetylase 1 (HDAC1) through RRM1 and RRM2 domain, which is known to contain two acetylation sites TDP-43 major, K142 and K192. In addition, we show that the TDP-43 is a direct transcriptional activator of CHOP promoter and this activity is regulated by acetylation.
Finally and most importantly, we observed both in cell culture and in Drosophila that HDCA1 reduced rate (genome inactivation or siRNA) or treatment with an inhibitor of pan-HDAC exert a protective role against WT or pathological mutant toxicity of TDP-43, show TDP-43 acetylation as a potential new therapeutic target.
HDAC inhibitory efficacy in neurodegeneration has long been debated, but future investigations warranted in this area. Selection of more specific HDAC inhibitor is still a promising option for the protection of neurons mainly because HDAC1 emerged as a downstream target both TDP- 43 and FUS gene-related ALS.
PLANT NATRIURETIC PEPTIDE A and Its Putative Receptor PNP-R2 Antagonize Salicylic Acid-mediated Signaling and Cell Death.
Plant stress hormone salicylic acid (SA) participated in local and systemic acquired resistance, which ultimately leads to a whole plant resistance to pathogens. However, if the SA-mediated signal is not precisely controlled, plants subject to fitness related defense costs such as growth inhibition and cell death.
Despite its importance, to date, only a few components counteracting stress responses SA-prime have been identified in Arabidopsis (Arabidopsis thaliana).
This includes other plant hormones such as jasmonic acid and abscisic acid, and proteins such as simulation LESIONS DISEASE 1 (LSD1), a transcription coregulator. Here, we describe PLANT A natriuretic peptide (PNP-A), functional analog for vertebrate atrial natriuretic peptide, which appears to antagonize the stress response SA-mediated plant. While the loss of the PNP-A potentiate SA-mediated signaling, synthetic exogenous application PNP-A or excess of PNP-A were significantly compromise SA-primed immune response.
In addition, we identify the plasma membrane-localized receptor-like protein, PNP-R2, which interacts with the PNP-A and is required to start the PNP-A-mediated intracellular signaling. In short, our work identifies peptides and cognate receptor is suspected as well counteracting SA-mediated signaling and cell death in Arabidopsis SA-primed.